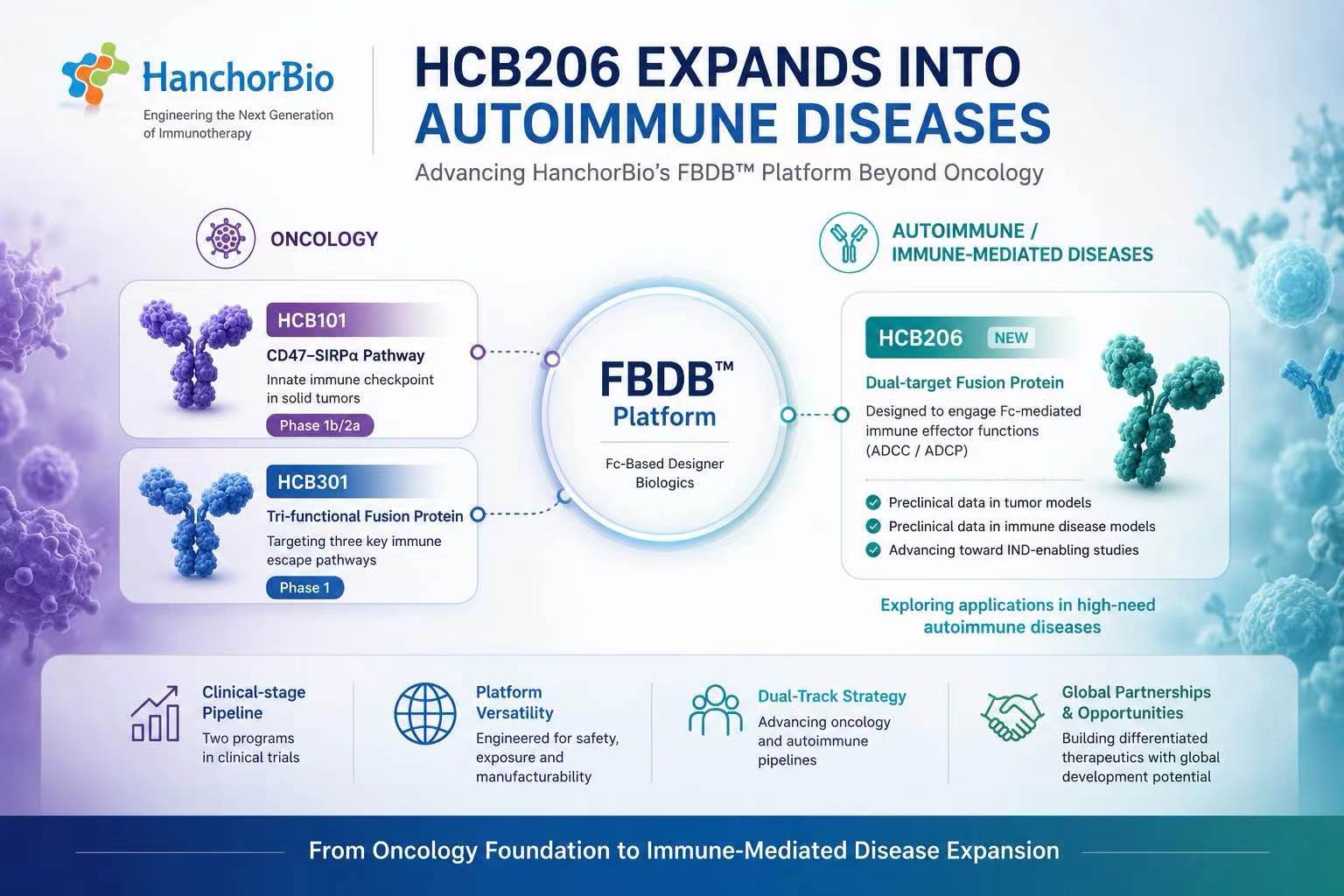
Our Possibility
我們的可能性
研發專案
8
+
專利申請
100
+
進入臨床IND階段
最快
2.3
年
2024年的研發投資金額
7.89
億元

FBDB™
技術平台的核心優勢
結合先天性及適應性免疫系統 多管齊下攻擊腫瘤細胞
阻斷 SIRPα / CD47 訊號傳遞
阻斷 PD-1 / PD-L1訊號傳遞
改善腫瘤微環境或進一步激活巨噬細胞/T細胞


News
最新消息

JOIN OUR TEAM